The therapeutic effect of anti-HER2/neu antibody depends on both innate and adaptive immunity. Trastuzumab increases HER2 uptake and cross-presentation by dendritic cells. Efficacy and safety of pembrolizumab or pembrolizumab plus chemotherapy vs chemotherapy alone for patients with first-line, advanced gastric cancer: the KEYNOTE-062 phase 3 randomized clinical trial. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): a phase 3, open-label, randomised controlled trial. Integrated genomic characterization of oesophageal carcinoma. Gastric Cancer 18, 476–484 (2015).Ĭancer Genome Atlas Research Network. HER2 screening data from ToGA: targeting HER2 in gastric and gastroesophageal junction cancer. Comprehensive molecular characterization of gastric adenocarcinoma. Access to genetic or exploratory biomarker data requires a detailed statistical analysis plan that is collaboratively developed by the requestor and MSD subject matter experts after approval of the statistical analysis plan and execution of a data-sharing agreement, MSD will either perform the proposed analyses and share the results with the requestor or will construct biomarker covariates and add them to a file with clinical data that is uploaded to a SAS portal so that the requestor can perform the proposed analyses.Ĭancer Genome Atlas Research Network. If the request is declined, it will be communicated to the investigator. 
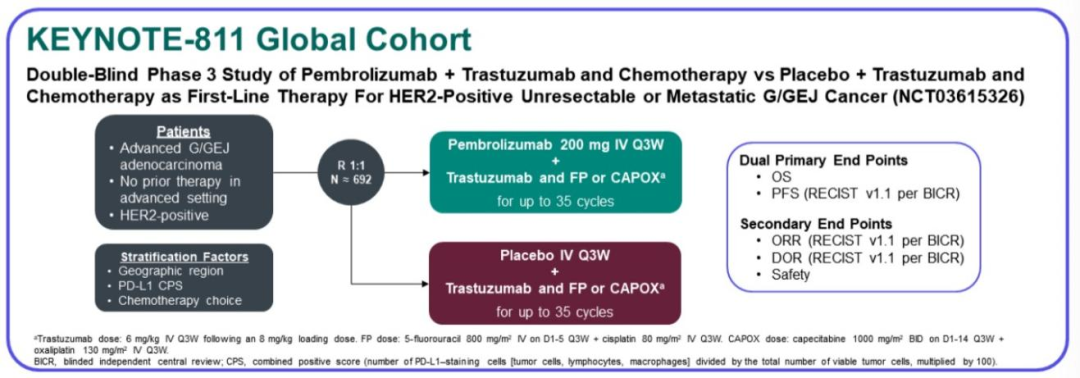
There are circumstances that may prevent MSD from sharing requested data, including country or region-specific regulations. Data will be made available for request after product approval in the US and EU or after product development is discontinued an exception will be made for this ongoing trial such that data will be made available for request after the protocol-specified primary endpoint analyses have been reported. In line with data privacy legislation, submitters of approved requests must enter into a standard data-sharing agreement with MSD before data access is granted. Feasible requests will be reviewed by a committee of MSD subject matter experts to assess the scientific validity of the request and the qualifications of the requestors. The MSD data-sharing documentation ( ) outlines the process and requirements for submitting a data request. MSD is also obligated to protect the rights and privacy of trial participants and, as such, has a procedure in place for evaluating and fulfilling requests for sharing company clinical trial data with qualified external scientific researchers. Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, NJ, USA (MSD) is committed to providing qualified scientific researchers access to anonymized data and clinical study reports from the company’s clinical trials for the purpose of conducting legitimate scientific research. We show that adding pembrolizumab to trastuzumab and chemotherapy markedly reduces tumour size, induces complete responses in some participants, and significantly improves objective response rate. Here we describe results of the protocol-specified first interim analysis of the randomized, double-blind, placebo-controlled phase III KEYNOTE-811 study of pembrolizumab plus trastuzumab and chemotherapy for unresectable or metastatic, HER2-positive gastric or gastro-oesophageal junction adenocarcinoma 22 (, NCT03615326). Although adding the anti-programmed death 1 (PD-1) antibody pembrolizumab to chemotherapy does not significantly improve efficacy in advanced HER2-negative gastric cancer 5, there are preclinical 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19 and clinical 20, 21 rationales for adding pembrolizumab in HER2-positive disease.  
More than a decade ago, combination therapy with the anti-HER2 antibody trastuzumab and chemotherapy became the standard first-line treatment for patients with these types of tumours 4. Human epidermal growth factor receptor 2 ( HER2, also known as ERBB2) amplification or overexpression occurs in approximately 20% of advanced gastric or gastro-oesophageal junction adenocarcinomas 1, 2, 3.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
